Abstract
Authors: Amit Bhattacharya, Ji-young Ock, Tao Wang, J. T. Bamford, Rachel A. Segalman, Sheng Dai, Alexei Sokolov, Xi Chen, and Raphaële Clément
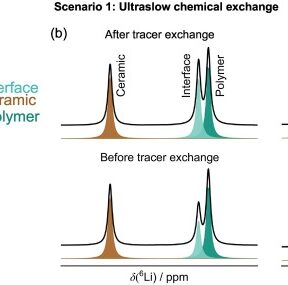
Abstract: Polymer-ceramic composites offer a path to enhance the transport and mechanical properties of solid electrolytes. However, an in-depth understanding of the extent and role of ion transport along and across polymer-ceramic interfaces in these systems is lacking. We have recently shown that Li-conducting Li0.11Na0.24K0.02La0.43TiO2.82 (LMTO) nanorods can be prepared by a molten flux method, and the addition of 30–50 weight (wt.)% LMTO to a bis[(trifluoromethyl)sulfonyl]imide-vinyl ethylene carbonate-based single-ion conducting (SIC) polymer electrolyte leads to a two-fold enhancement in Li-ion conductivity, from 1.4 to 3.0 × 10−5 S/cm at 30 °C. In the present study, we use NMR methods to identify the Li-ion transport pathways and determine the timescale of chemical exchange between the SIC polymer and LMTO ceramic components. Tracer exchange NMR indicates preferential transport through the polymer or polymer-interfacial regions, and exchange spectroscopy (EXSY) and a new isotope exchange method reveal negligible Li exchange between the SIC polymer and LMTO ceramic up to several days. Here, LMTO nanorods act as a passive filler. Our results further highlight that significant (e.g., 10- or 100-fold) conductivity enhancements in composite electrolytes can only be achieved 1) with ionically-conductive fillers, and 2) when both the ceramic and polymer components actively participate in long-range transport. For this, fast interfacial ion exchange is needed. This leads us to introduce a critical interfacial ion exchange time to evaluate whether a filler actively contributes to conduction in a composite electrolyte, and screen for promising polymer-ceramic pairings to accelerate the development of high conductivity solid electrolytes.