Abstract
Authors: Vaidyanathan Sethuraman, Ji-young Ock, Michelle L. Lehmann, Andrew Ullman, Rajeev Kumar, Xi Chelsea Chen, and Lei Cheng
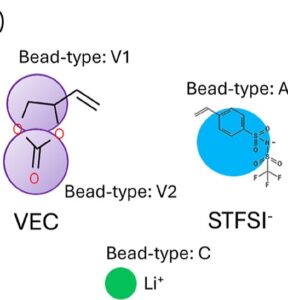
Abstract: We use a combination of experiments and coarse-grained molecular dynamics simulations to elucidate the structure–property relationships in polymer electrolytes obtained by the copolymerization of poly(vinyl ethylene carbonate─lithium styrene bis(trifluoromethanesulfonyl)imide) or p(VEC-LiSTFSI). Experiments show that the conductivity reduces with increasing anion (i.e., STFSI) fraction on the chain, and the cation transference number (t+) is found to be dependent on the anion fraction. Furthermore, a significant fraction of unpolymerized VEC monomers are observed. Since it is inherently difficult to experimentally control the chain architecture and the amount of unpolymerized VEC in these systems, we perform coarse-grained molecular dynamics simulations on model polymer systems with different chain architectures to mimic the plausible experimental systems. Specifically, we look at the differences in transference numbers arising from (i) a random copolymer of VEC and STFSI monomers; (ii) a blend of VEC-STFSI copolymer with VEC monomers; and (iii) a ternary blend of the VEC homopolymer, STFSI homopolymers, and VEC monomers. The ternary blend model demonstrates the closest resemblance with the experimental transference numbers and diffusivities. The lithium diffusivity obtained from the coarse-grained models with VEC monomers (plasticizers) is about 1.5 times that of the model without VEC monomers, showing that the plasticizing effect of VEC monomers is modest. We rationalize the experimental observations based on aggregate and cluster analyses obtained from molecular simulations. This work reveals that polymer electrolyte chain architecture and plasticizers can critically influence the transport properties, and these parameters should be considered when designing single ion conducting polymeric electrolytes.